Revision of plan for integration of medical device businesses
Kuraray Medical (KM), wholly owned subsidiary of Kuraray, and Asahi Kasei Medical (AM), wholly owned subsidiary of Asahi Kasei Pharma, have revised their plan for the integration of medical device businesses as announced on December 14, 2006.
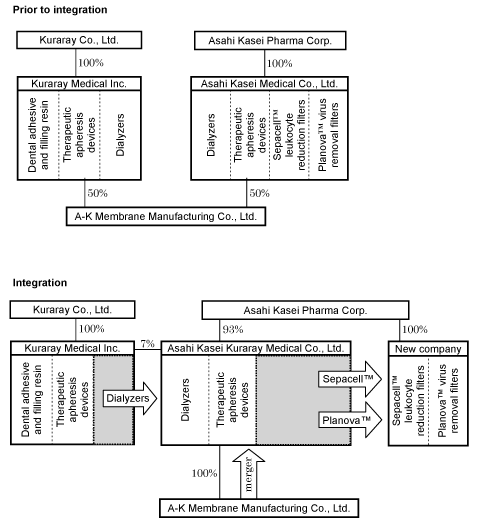
Businesses subject to integration on October 1, 2007
| As previously announced | Dialyzers and therapeutic apheresis devices of AM and KM. |
|---|---|
| As revised | Dialyzers and therapeutic apheresis devices of AM and dialyzers of KM. |
Proportion of ownership of the integrated company
| As previously announced | 85% by Asahi Kasei Pharma, 15% by Kuraray Medical |
|---|---|
| As revised | 93% by Asahi Kasei Pharma, 7% by Kuraray Medical |
Reason for revision
In the course of prior consultation with the Japan Fair Trade Commission (JFTC), it became apparent that a response from the JFTC to the effect that there would be no concern of restraint on competition in the domestic market for therapeutic apheresis devices would not be forthcoming soon enough to enable the planned business integration to proceed on schedule. AM and KM therefore opted to proceed first with an integration only of their dialyzer businesses. The revision of the proportion of ownership of the integrated company reflects valuation based on the revised scope of businesses subject to integration.
Outlook
AM produces devices based on polysulfone hollow-fiber membrane technology, the global standard for high-performance clearance of low-molecular-weight proteins, and devices based on cellulose hollow-fiber technology, while KM produces devices based on Eval® ethylene-vinyl alcohol copolymer (EVOH) hollow-fiber technology, featuring outstanding biocompatibility and enabling mild and gentle therapy for older patients and those just beginning treatment. The integration of the two companies’ hollow-fiber technology and know-how will accelerate the development of next-generation devices which offer greater safety and efficacy, enabling the integrated operation to enjoy a solid position as a global leader in medical devices, well placed for expansion and growth.
Integration process
The Sepacell™ leukocyte reduction filter and Planova™ virus removal filter businesses of AM will be transferred to a newly established company. AM, retaining its dialyzer and therapeutic apheresis device businesses, will be renamed Asahi Kasei Kuraray Medical Co., Ltd. The dialyzer businesses of KM will be transferred to Asahi Kasei Kuraray Medical. KM will retain its dental adhesive and filling resin business and its therapeutic apheresis device business. A-K Membrane Manufacturing Co., Ltd., joint venture of AM and KM for the production of Eval® hollow-fiber membrane for medical devices, will become a wholly owned subsidiary of Asahi Kasei Kuraray Medical, and then merged with that company.
Schematic representation of the business integration
(percentages indicate ownership)
Profile of the integrated business
| Company name | Asahi Kasei Kuraray Medical Co., Ltd. |
|---|---|
| President | To be named from Asahi Kasei Medical |
| Scheduled start of operation | October 1, 2007 |
| Paid-in capital | ¥800 million (tentative) |
| Shareholding | 93% by Asahi Kasei Pharma, 7% by Kuraray Medical |
| Head office | Tokyo, Japan |
| Business line | Development, production, and sale of dialyzers and therapeutic apheresis products |
| Production sites | Nobeoka, Miyazaki, Japan; Oita, Japan; Hangzhou, Zhejiang, China |
| Sales offices | Japan, US, EU, China, Korea |
| Employees | Approx. 1,400 |
| FY 2007 sales forecast | ¥40 billion (annualized) |
| FY 2010 sales forecast | ¥48 billion |
Kuraray Medical corporate profile
| President | Hideo Horii |
|---|---|
| Establishment | June 2001 |
| Paid-in capital | ¥2.5 billion |
| Shareholding | 100% by Kuraray Co., Ltd. |
| Head office | Tokyo, Japan |
| Main products | Dialyzers, therapeutic apheresis products, dental adhesive and filling resin |
| Production facilities | Kurashiki - production of hollow-fiber dialyzer membrane, dental adhesive, and dental filling resin |
| Employees | Approx. 300 (as of Mar. 31, 2007) |
| Sales | ¥11.8 billion (year ended March 2007) |
Asahi Kasei Medical corporate profile
| President | Yasuyuki Yoshida |
|---|---|
| Establishment | July 1974 |
| Paid-in capital | ¥800 million |
| Shareholding | 100% by Asahi Kasei Pharma |
| Head office | Tokyo, Japan |
| Main products | Dialyzers, therapeutic apheresis products, leukoreduction filters for transfusion, virus removal filters |
| Production facilities | Nobeoka - production of hollow-fiber dialyzer membrane and other products Oita - assembly of dialyzers and blood filters China - assembly of dialyzers |
| Employees | Approx. 1,700 (as of Mar. 31, 2007) |
| Sales | ¥43.1 billion (year ended March 2007) |
